
Early improvements with long-term impact in Alagille syndrome (ALGS)

pivotal study of its kind
The ICONIC study assessed efficacy and safety of treatment with LIVMARLI in
patients ≥1 year old with cholestatic pruritus associated with ALGS1,2
Cholestatic pruritus responses to LIVMARLI were assessed through approximately 4 years.
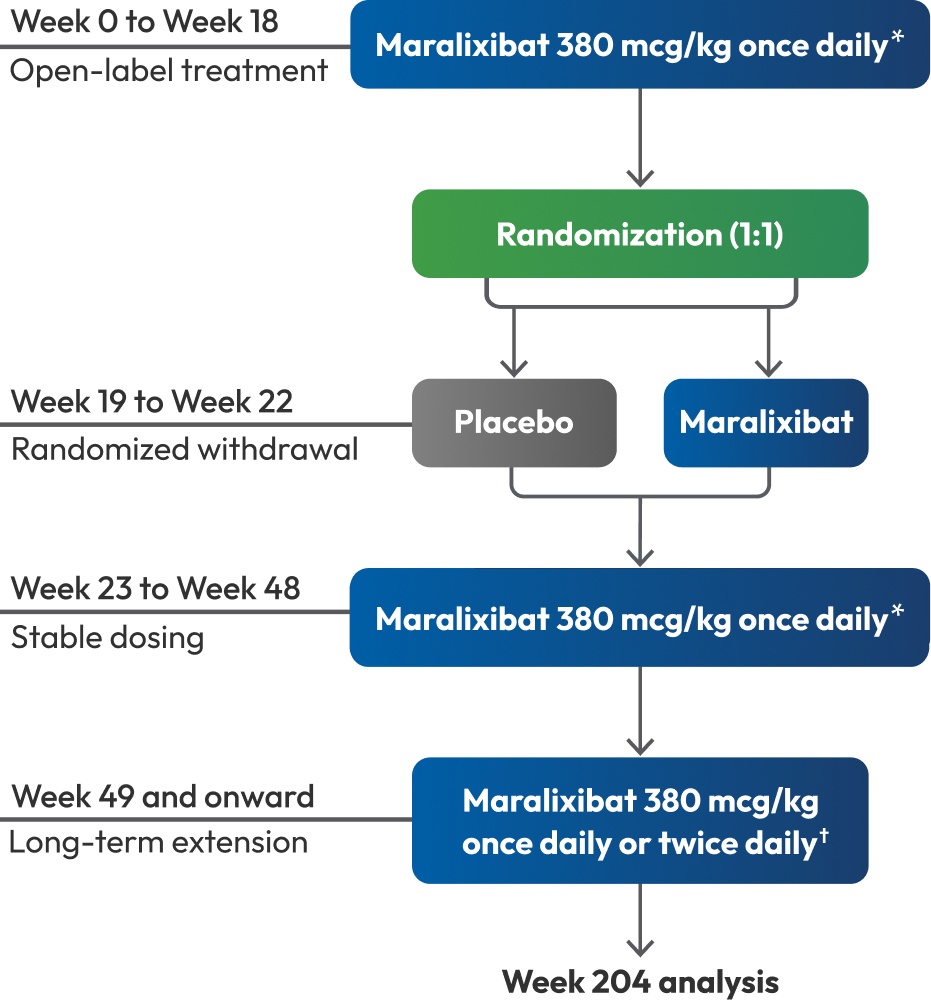
- * Included a 6-week dose escalation period for all participants during the first 6 weeks of the open-label treatment period and for participants who received placebo during the randomized withdrawal design.
- † Twice-per-day dosing was allowed after Week 100. The approved dosage of LIVMARLI is 380 mcg/kg once daily.
CSS=Clinician Scratch Scale; ItchRO(Obs)=Itch Reported Outcome for Observer; SD=standard deviation.
- ‡ Average ItchRO(Obs) scores are based on the 7 days prior to baseline visit.
Primary endpoint: Change in sBA levels during the randomized withdrawal period in participants who previously achieved an sBA reduction of at least 50% from baseline to Week 12 or Week 18.1
Key pruritus endpoints: Difference in pruritus severity (measured by the weekly morning average ItchRO[Obs] score) during the randomized withdrawal and the change from baseline to Week 48.1
Key secondary endpoints: sBA level and ItchRO(Pt) scores in all study participants from baseline to Weeks 18 and 48.1
ItchRO(Obs)=Itch Reported Outcome for Observer; ItchRO(Pt)=Itch Reported Outcome for Patient; sBA=serum bile acid.
Significant improvements in cholestatic pruritus from baseline were seen at the very first assessment (Week 3), with the full effect achieved by Week 18 and maintained through 1 year with once-daily LIVMARLI (P<0.0001).3,4
- Cholestatic pruritus was assessed each day, in the morning and evening, using the ItchRO scale—a validated tool designed to assess the impact of cholestatic pruritus in people with cholestatic liver disease, including ALGS. The ItchRO score is a 0–4 scale, where 0 is none, 1 is mild, 2 is moderate, 3 is severe, and 4 is very severe3,4
- “Clinically meaningful” was defined as ≥1.0 ItchRO(Obs) improvement vs baseline
Efficacy demonstrated in patients who were on background pruritus
therapies, including ursodeoxycholic acid and rifampicin1
BL=baseline; ItchRO=Itch Reported Outcome; ItchRO(Obs)=Itch Reported Outcome for Observer; ItchRO(Pt)=Itch Reported Outcome (Patient); sBA=serum bile acid; SE=standard error.
- * ItchRO(Obs) was completed by caregivers and was the basis for the key pruritus endpoint. The ItchRO(Pt) was completed independently by participants aged 9 years or older and with caregiver assistance for participants aged 5 to 8 years.3,4
- † Change from baseline, P<0.0001.3,4
- ‡ Included an initial 6-week dose escalation for participants previously receiving placebo.3,4
-
The MERGE study followed up with all patients from ICONIC, IMAGINE, and IMAGINE-II to assess the long-term durability of response to LIVMARLI.5
- Subsequent data are from an analysis of patients who remained on LIVMARLI, including 23 patients who remained on treatment for 7 years. Some patients in this analysis continued treatment with LIVMARLI due to a perceived benefit, even though they did not meet the criteria for a cholestatic pruritus response5
-
Change from baseline was assessed by comparing median (Q1, Q3) values from enrollment in the initial trial (ie, ICONIC, IMAGO, or ITCH) with data from the MERGE visit that most closely aligned with an annual visit.5
- *IMAGINE and IMAGINE-II were extension studies of IMAGO and ITCH, respectively.
This analysis assessed the impact of treatment response to LIVMARLI on patients with ALGS health-related quality of life (HRQoL) and used data from the pivotal trial, ICONIC. The Pediatric Quality of Life Inventory (PedsQL) Multidimensional Fatigue Scale was measured using a caregiver proxy report at baseline and Weeks 18, 22, and 48 that was collected prospectively during the study and analyzed retrospectively. As these scales were not specifically developed for patients with ALGS, a subset of individual items from the HRQoL scales deemed most relevant was independently selected by clinical experts for assessment with treatment response.6
During the first year of treatment, 74% (n/N=20/27) of patients treated with LIVMARLI were considered responders.6
- Responders were defined as patients who had a 1-point ItchRO(Obs) improvement from baseline to Week 486
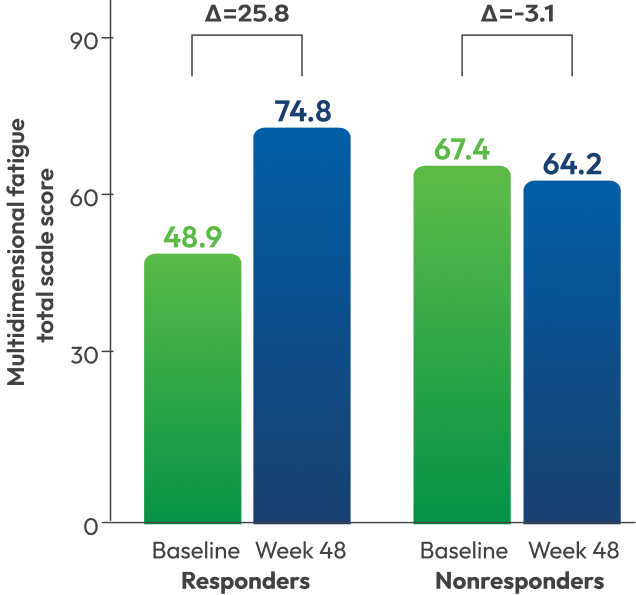
- The mean (standard deviation [SD]) change in the Multidimensional Fatigue Scale total score was +25.8 (23.0) for responders vs –3.1 (19.8) for nonresponders6
- The data were available in 21 patients at baseline and Week 48 and 6 patients had missing data at Week 486
fatigue while being treated with LIVMARLI.6
Limitations6
- The PedsQL Multidimensional Fatigue Scale has not been optimized for pediatric patients with cholestatic diseases
- This analysis was limited by small sample sizes, in some cases due to missing data, meaning some of the analyses may have been underpowered. This study did not adjust for multiplicity
- Sleep-related items may have improved because reductions in pruritus results in fewer disruptions and a better quality of sleep
- Given these limitations, results should be interpreted with caution
*A higher value in the multidimensional fatigue total score represents a positive response.
Assessed multiple facets of fatigue6
LIVMARLI responders experienced improvements in 5 sleep-related assessments of the PedsQL Multidimensional Fatigue Scale compared with nonresponders, including:
-
Feeling tired
-
Sleeping a lot
-
Difficulty sleeping through the night
-
Feeling tired upon waking
-
Taking a lot of naps
Additional parameters assessed in the PedsQL Multidimensional Fatigue Scale included “difficulty keeping his or her attention” and “resting a lot,” but no meaningful results were observed.6

Téa struggled with persistent itching and disrupted sleep until she started LIVMARLI
"After about 14 days on LIVMARLI, we noticed a change in her itching."
–Michelle, Téa's mom
Not all patients taking LIVMARLI and their caregivers will have the same experience.
Michelle: We first learned about LIVMARLI through the clinical trials. We knew that there was something in the works for itching, which we were really excited about. Téa started LIVMARLI in February of 2022, when she was 8 years old. After about 14 days on LIVMARLI, we noticed a drastic change in her itching. Her first night, we knew that LIVMARLI was working for her. In the middle of the night, I didn't hear her itching. She was snoring. It was so funny to me, but also very sad to know that it took 8 years for her to sleep all night without sitting up and itching in the middle of the night. And in the morning, she ran into my room and she's like, "Mama, I slept so good."