
How LIVMARLI works

In the treatment of Alagille syndrome (ALGS) and progressive familial intrahepatic cholestasis (PFIC)
LIVMARLI targets cholestatic pruritus
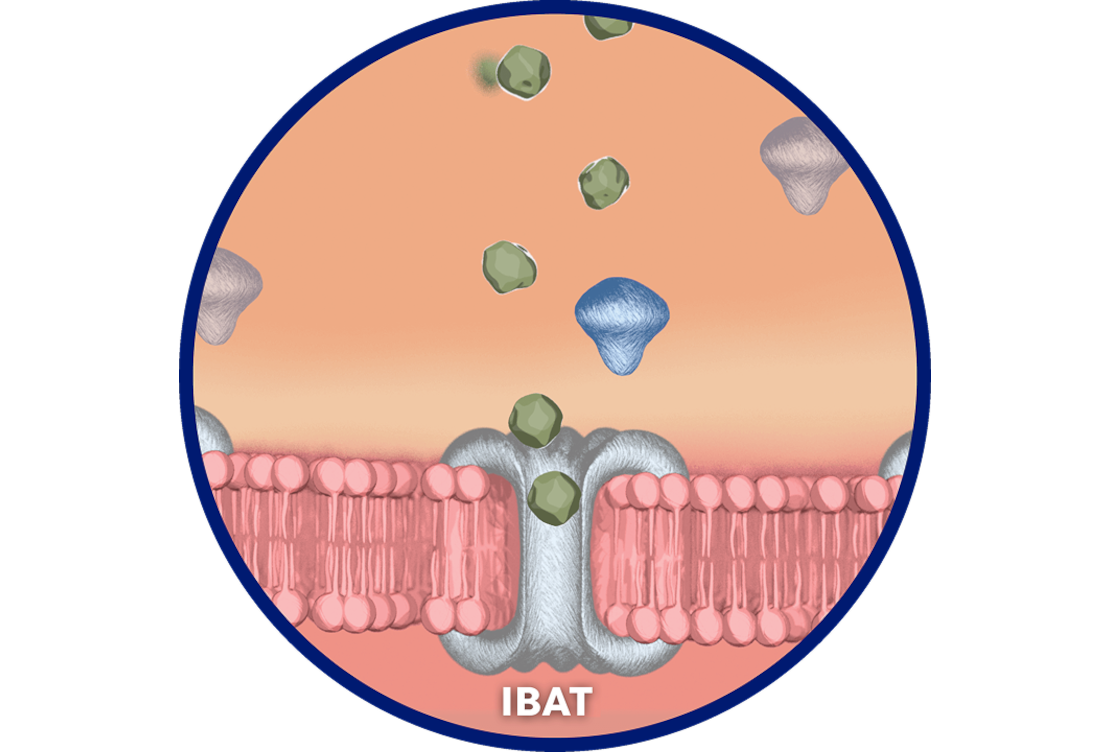

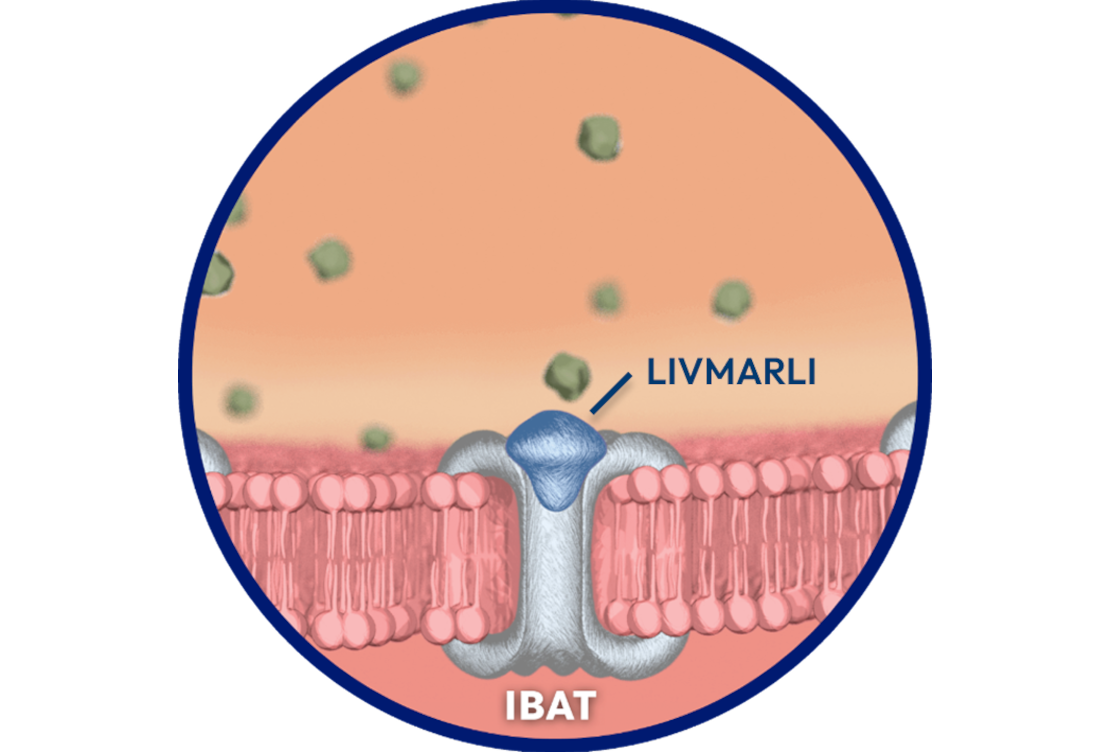
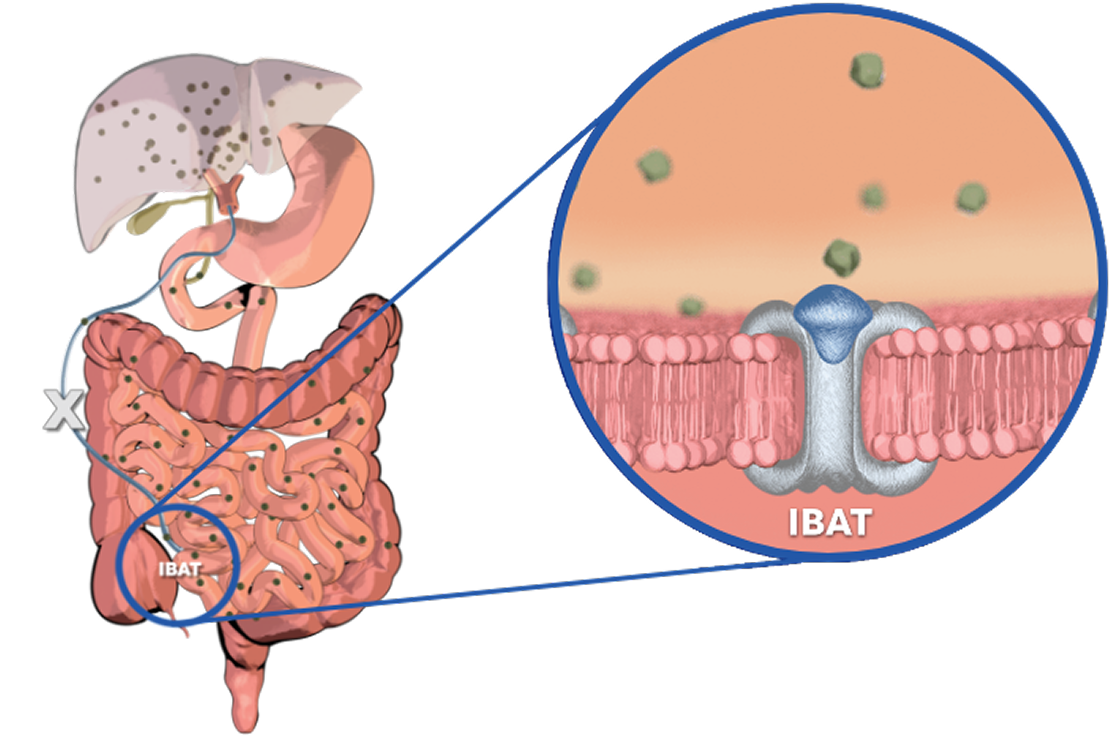
LIVMARLI helps reduce bile acid buildup.


LIVMARLI interrupts recirculation of bile acids to the liver
and increases their fecal excretion. It reduces bile acid levels in the body (as measured by serum bile acids [sBAs]), with minimal systemic absorption.1-3

LIVMARLI decreases reuptake of bile salts
as observed by a decrease in sBA.1-3
The complete mechanism by which LIVMARLI improves cholestatic pruritus is unknown, but it may involve inhibition of the IBAT.1
Prescription access and support
Mirum Access Plus supports you and your patients through payer approval and beyond.
References:
1. LIVMARLI® (maralixibat). Prescribing Information. Mirum Pharmaceuticals, Inc.
2. Gonzales E, Hardikar W, Stormon M, et al. Efficacy and safety of maralixibat treatment in patients with Alagille syndrome and cholestatic pruritus (ICONIC): a randomised phase 2 study. Lancet. 2021;398(10311):1581-1592. doi:10.1016/S0140-6736(21)01256-3
3. Martin P, Apostol G, Smith W, Jennings L, Vig P. Dose-dependent fecal bile acid excretion with apical sodium-dependent bile acid transporter inhibitors maralixibat and volixibat in a dose-ranging phase 1 study in overweight and obese adults. Poster presented at: American Association for the Study of Liver Diseases: The Liver Meeting; November 8-12, 2019; Boston, MA.