Tyler, real patient living with Alagille syndrome and his mom, Terisa
Individual experiences may vary.



Tyler, real patient living with Alagille syndrome and his mom, Terisa
“It’s fine”
Are your patients really fine? Many patients with Alagille syndrome and their families may minimize the symptoms of cholestatic pruritus. Hear from these real patients and their families about how they realized they weren’t fine and what they did to address it. Their experiences may inspire you to inquire further with your patients.
Because any level of cholestatic pruritus deserves attention.
Individual experiences may vary.








People say,
“I’m fine”
Some facts
around fine
Are your conversations about cholestatic pruritus only skimming the surface?
In one survey of patients with Alagille syndrome,
Nearly 30%
said they talked to their doctor about cholestatic pruritus, but not in detail.2
However,
~20%
said cholestatic pruritus was not discussed at all.2
“We really didn’t want to acknowledge the itching. We just found ways around it. We would keep the house cool. We would wash her sheets every day. She wasn’t fine.”
Téa | 10 years old
Real patient living with Alagille syndrome and her mom, Michelle
Individual experiences may vary.
“He’d shuffle his feet, kick his legs—what baby doesn’t? Looking back now, it wasn’t typical newborn behavior. He wasn’t fine.”
Finley | 3 years old
Real patient living with Alagille syndrome and his parents, Jay and Dani
Individual experiences may vary.
Patients and caregivers may normalize symptoms
The signs and symptoms of cholestatic pruritus can appear in many ways and may be difficult to connect to Alagille syndrome.
- Seasonal changes
- Possible irritants, like laundry soap
- Skin conditions that may cause itch
- Allergies
- Symptoms were mild compared with those experienced by others with cholestatic pruritus in Alagille syndrome
- It was just a “flare-up”
- They could wait it out
- Nothing more could be done
Patients may not be comfortable bringing up cholestatic pruritus on their own
of patients with chronic illness
find it difficult to share their feelings with their physician.9“I learned to fight the urge to scratch in public. Before I’d go anywhere I would take time out of my day to scratch. Looking back now,
I was not fine.”
Emma | 19 years old
Real patient living with Alagille syndrome
Individual experiences may vary.
Hear their stories
surrounding the fines








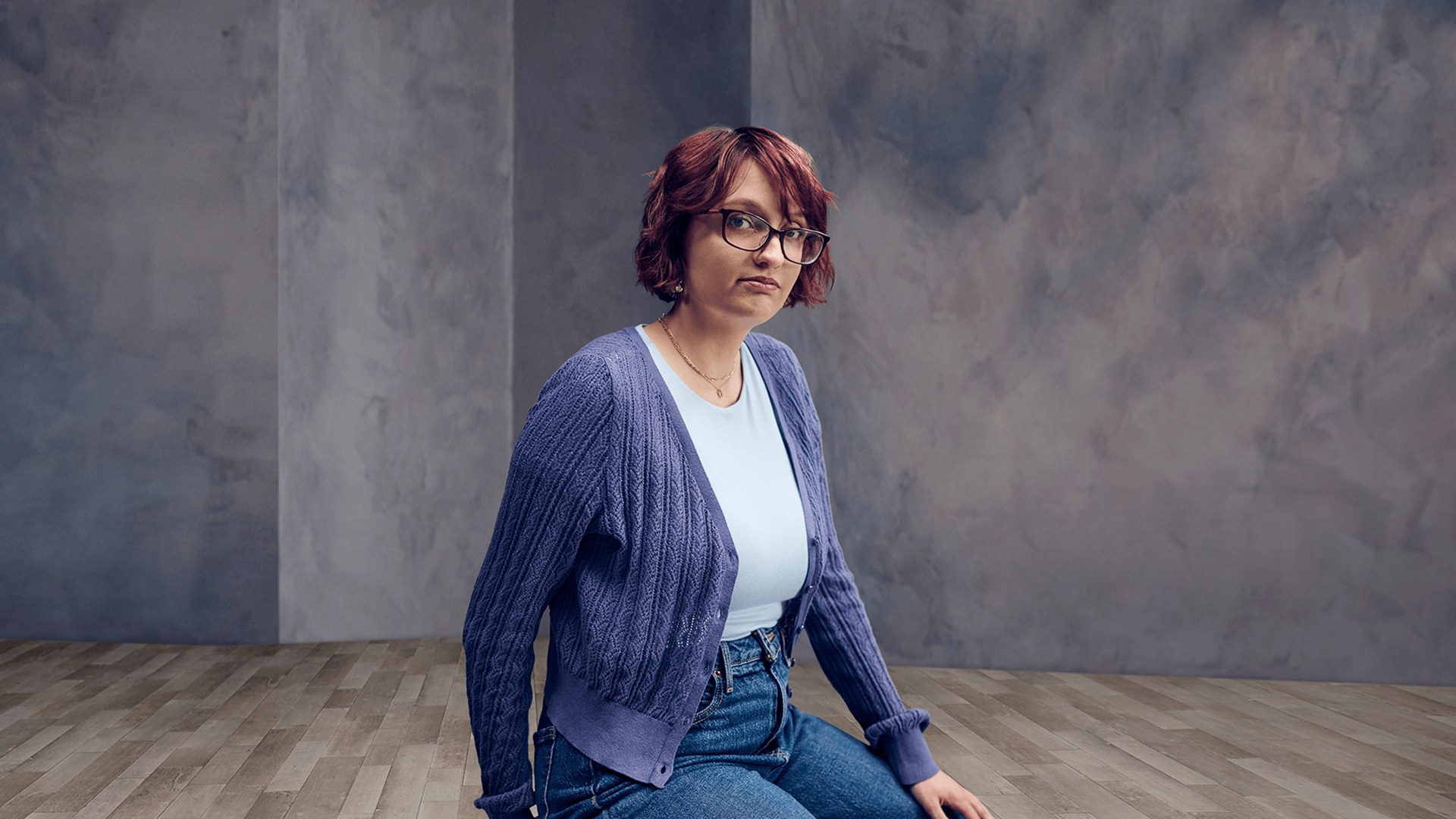



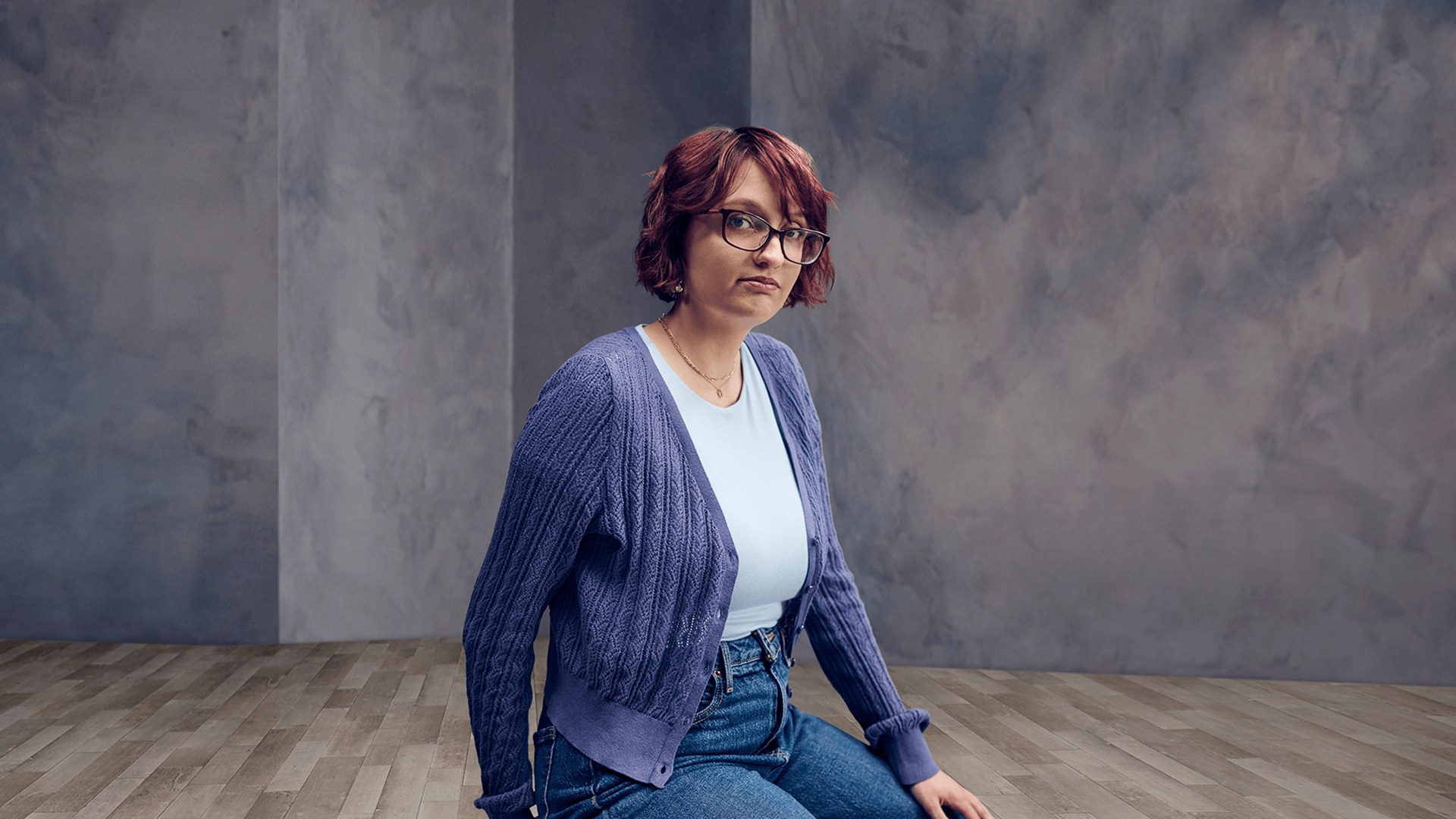





Their story
Meet patients living with Alagille syndrome who normalized the symptoms of cholestatic pruritus.
Emma’s story
Meet Emma, who would schedule time to scratch before leaving the house.
Téa’s story
Meet Téa and her mom, Michelle, who found ways to work around cholestatic pruritus.
Abby’s story
Meet Abby, who just accepted cholestatic pruritus as part of her life because she did not know what else could be done.
Finley’s story
Meet Jay and Dani, parents to Finley, who thought his expressions of cholestatic pruritus were normal newborn behavior.
Tyler’s story
Meet Tyler and his mom, Terisa, who mistook his cholestatic pruritus for allergies or changes in weather.
[00:00:00]
[pause 00:00:00]
[music]
Emma: I learned to fight the urge to scratch in public. Actually, before I’d go anywhere, I would just scratch while I was still home. Eventually, though, I just broke down. I was not fine.
Jay: We tried consoling him, but nothing works. In [00:00:30] hindsight, it wasn’t typical newborn behavior. Something else was wrong, and he wasn’t fine.
Terisa: I tried blaming his itch on something else. I told myself, “It’s fine, maybe he just has allergies.” We learned to live with it because that’s what was normal for us. We didn’t know any different, but I know now he was far from fine.
Abby: I didn’t realize how bad the itch was until I found relief. Looking back, I was scared to talk about my cholestatic pruritus because [00:01:00] I didn’t know what my future looked like. Now I understand, I wasn’t fine.
Michelle: It wasn’t until Téa started taking LIVMARLI that we realized how bad her itch had been. Scratching through the night should not be the norm. Looking back, she wasn’t fine. Are you reading between the fines?
Voiceover 1: Any level of cholestatic pruritus in Alagille syndrome deserves to be addressed. Find out how you can help your patients talk about their symptoms.
Voiceover 2:
INDICATION
[00:01:30] LIVMARLI is indicated for the treatment of cholestatic pruritus in patients who are 3 months of age and older with Alagille syndrome.
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
Contraindicated in patients with prior or active hepatic decompensation events (eg, variceal hemorrhage, ascites, or hepatic encephalopathy).
WARNINGS AND PRECAUTIONS
Hepatotoxicity: Treatment-emergent elevations or worsening of liver tests have occurred. Obtain baseline liver tests [00:02:00] and monitor during treatment; monitor also for liver-related adverse reactions and signs of hepatic decompensation. Dose reduction or treatment interruption may be considered if abnormalities occur. Permanently discontinue if persistent liver test abnormalities, clinical hepatitis upon rechallenge, or hepatic decompensation occur.
Gastrointestinal Adverse Reactions: Diarrhea and abdominal pain were reported as the most common adverse reactions in patients treated with LIVMARLI. [00:02:30] Consider reducing the dosage or interrupting treatment if a patient experiences persistent diarrhea or abdominal pain.
Fat-Soluble Vitamin (FSV) Deficiency: Obtain baseline levels and monitor during treatment. Supplement vitamins if deficiency is observed. Consider interrupting LIVMARLI if bone fractures or bleeding occur.
Risk of Propylene Glycol Toxicity (Pediatric Patients Less Than 5 Years of Age): Total daily intake of propylene glycol should be considered for managing [00:03:00] the risk of propylene glycol toxicity. Monitor patients for signs of propylene glycol toxicity. Discontinue LIVMARLI if toxicity is suspected.
[music]
[00:03:14] [END OF AUDIO]
Emma: I learned to fight the urge to scratch in public. Before I’d go anywhere, I would take time out of my day to scratch. When people had asked about my itching, though, I just said I was fine because I felt helpless at the time. They couldn’t help me and I couldn’t help myself, so why even bring it up? Looking back now, though, I was not fine.
Voiceover 1: Any level of cholestatic pruritus in Alagille syndrome deserves to be addressed. Find out how you can help your patients talk about their symptoms [00:00:30].
Voiceover 2:
INDICATION
LIVMARLI is indicated for the treatment of cholestatic pruritus in patients who are 3 months of age and older with Alagille syndrome.
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
Contraindicated in patients with prior or active hepatic decompensation events (eg, variceal hemorrhage, ascites, or hepatic encephalopathy).
WARNINGS AND PRECAUTIONS
Hepatotoxicity: Treatment-emergent elevations [00:01:00] or worsening of liver tests have occurred. Obtain baseline liver tests and monitor during treatment; monitor also for liver-related adverse reactions and signs of hepatic decompensation. Dose reduction or treatment interruption may be considered if abnormalities occur. Permanently discontinue if persistent liver test abnormalities, clinical hepatitis upon rechallenge, or hepatic decompensation occur.
Gastrointestinal Adverse Reactions: Diarrhea and abdominal pain were reported as the most common adverse reactions [00:01:30] in patients treated with LIVMARLI. Consider reducing the dosage or interrupting treatment if a patient experiences persistent diarrhea or abdominal pain.
Fat-Soluble Vitamin (FSV) Deficiency: Obtain baseline levels and monitor during treatment. Supplement vitamins if deficiency is observed. Consider interrupting LIVMARLI if bone fractures or bleeding occur.
Risk of Propylene Glycol Toxicity (Pediatric Patients Less Than 5 Years of Age): Total daily intake [00:02:00] of propylene glycol should be considered for managing the risk of propylene glycol toxicity. Monitor patients for signs of propylene glycol toxicity. Discontinue LIVMARLI if toxicity is suspected.
[00:02:17] [END OF AUDIO]
Michelle: We really didn’t want to acknowledge the itching. We just found ways around it. We would keep the house cool. We would wash her sheets every day. And then, when Téa started taking LIVMARLI, we really realized that she hadn’t been fine.
Voiceover 1: Any level of cholestatic pruritus in Alagille syndrome deserves to be addressed. Find out how you can help your patients talk about their symptoms.
Voiceover 2:
INDICATION
LIVMARLI is indicated for the treatment of cholestatic pruritus in patients who are 3 months of age and older [00:00:30] with Alagille syndrome.
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
Contraindicated in patients with prior or active hepatic decompensation events (eg, variceal hemorrhage, ascites, or hepatic encephalopathy).
WARNINGS AND PRECAUTIONS
Hepatotoxicity: Treatment-emergent elevations or worsening of liver tests have occurred. Obtain baseline liver tests and monitor during treatment; monitor also for liver-related adverse reactions [00:01:00] and signs of hepatic decompensation. Dose reduction or treatment interruption may be considered if abnormalities occur. Permanently discontinue if persistent liver test abnormalities, clinical hepatitis upon rechallenge, or hepatic decompensation occur.
Gastrointestinal Adverse Reactions: Diarrhea and abdominal pain were reported as the most common adverse reactions in patients treated with LIVMARLI. Consider reducing the dosage or interrupting treatment if a patient experiences persistent diarrhea [00:01:30] or abdominal pain.
Fat-Soluble Vitamin (FSV) Deficiency: Obtain baseline levels and monitor during treatment. Supplement vitamins if deficiency is observed. Consider interrupting LIVMARLI if bone fractures or bleeding occur.
Risk of Propylene Glycol Toxicity (Pediatric Patients Less Than 5 Years of Age): Total daily intake of propylene glycol should be considered for managing the risk of propylene glycol toxicity. Monitor patients for signs of propylene glycol toxicity [00:02:00]. Discontinue LIVMARLI if toxicity is suspected.
[00:02:09] [END OF AUDIO]
Abby: [00:00:00] I itched and rubbed my feet, and my friends used to think it was just who I was, and I had accepted it too. It wasn’t until I found relief that I realized how bad it was and how important it is to talk to your doctor. I was always scared to talk about my cholestatic pruritus, because I didn’t know what else could be done. Now I understand, I wasn’t fine.
Voiceover 1: Any level of cholestatic pruritus in Alagille syndrome deserves to be addressed. Find out how you can help your patients talk about their symptoms [00:00:30].
Voiceover 2:
INDICATION
LIVMARLI is indicated for the treatment of cholestatic pruritus in patients who are 3 months of age and older with Alagille syndrome.
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
Contraindicated in patients with prior or active hepatic decompensation events (eg, variceal hemorrhage, ascites, or hepatic encephalopathy).
WARNINGS AND PRECAUTIONS
Hepatotoxicity: Treatment-emergent elevations [00:01:00] or worsening of liver tests have occurred. Obtain baseline liver tests and monitor during treatment; monitor also for liver-related adverse reactions and signs of hepatic decompensation. Dose reduction or treatment interruption may be considered if abnormalities occur. Permanently discontinue if persistent liver test abnormalities, clinical hepatitis upon rechallenge, or hepatic decompensation occur.
Gastrointestinal Adverse Reactions: Diarrhea and abdominal pain were reported as the most common adverse reactions [00:01:30] in patients treated with LIVMARLI. Consider reducing the dosage or interrupting treatment if a patient experiences persistent diarrhea or abdominal pain.
Fat-Soluble Vitamin (FSV) Deficiency: Obtain baseline levels and monitor during treatment. Supplement vitamins if deficiency is observed. Consider interrupting LIVMARLI if bone fractures or bleeding occur.
Risk of Propylene Glycol Toxicity (Pediatric Patients Less Than 5 Years of Age): Total daily intake [00:02:00] of propylene glycol should be considered for managing the risk of propylene glycol toxicity. Monitor patients for signs of propylene glycol toxicity. Discontinue LIVMARLI if toxicity is suspected.
[00:02:17] [END OF AUDIO]
Dani: [00:00:00] We tried to console him and nothing worked. We’d give him a bottle but he wasn’t hungry. He’d rub against me. I just thought he was being affectionate. He’d shuffle his feet, kick his legs. What baby doesn’t? Looking back now, it wasn’t typical newborn behavior. He wasn’t fine.
Voiceover 1: Any level of cholestatic pruritus in Alagille syndrome deserves to be addressed [00:00:30]. Find out how you can help your patients talk about their symptoms.
Voiceover 2:
INDICATION
LIVMARLI is indicated for the treatment of cholestatic pruritus in patients who are 3 months of age and older with Alagille syndrome.
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
Contraindicated in patients with prior or active hepatic decompensation events (eg, variceal hemorrhage, ascites, or hepatic encephalopathy).
WARNINGS AND PRECAUTIONS
Hepatotoxicity: [00:01:00] Treatment-emergent elevations or worsening of liver tests have occurred. Obtain baseline liver tests and monitor during treatment; monitor also for liver-related adverse reactions and signs of hepatic decompensation. Dose reduction or treatment interruption may be considered if abnormalities occur. Permanently discontinue if persistent liver test abnormalities, clinical hepatitis upon rechallenge, or hepatic decompensation occur.
Gastrointestinal Adverse Reactions: Diarrhea and abdominal pain [00:01:30] were reported as the most common adverse reactions in patients treated with LIVMARLI. Consider reducing the dosage or interrupting treatment if a patient experiences persistent diarrhea or abdominal pain.
Fat-Soluble Vitamin (FSV) Deficiency: Obtain baseline levels and monitor during treatment. Supplement vitamins if deficiency is observed. Consider interrupting LIVMARLI if bone fractures or bleeding occur.
Risk of Propylene Glycol Toxicity (Pediatric Patients [00:02:00] Less Than 5 Years of Age): Total daily intake of propylene glycol should be considered for managing the risk of propylene glycol toxicity. Monitor patients for signs of propylene glycol toxicity. Discontinue LIVMARLI if toxicity is suspected.
[00:02:19] [END OF AUDIO]
Terisa: [00:00:00] He used to fidget a lot and I would tell myself, it’s fine. Maybe he just has allergies or maybe it’s the weather and everyone around us had downplayed it, so it became normal for us. When I did talk to his doctor, I felt he listened, but we did feel that we exhausted all the options of treatment for cholestatic pruritus, but we know now he was far from fine.
Voiceover 1: Any level of cholestatic pruritus in Alagille syndrome deserves to be addressed. Find out how you can help your patients [00:00:30] talk about their symptoms.
Voiceover 2:
INDICATION
LIVMARLI is indicated for the treatment of cholestatic pruritus in patients who are 3 months of age and older with Alagille syndrome.
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
Contraindicated in patients with prior or active hepatic decompensation events (eg, variceal hemorrhage, ascites, or hepatic encephalopathy).
WARNINGS AND PRECAUTIONS
Hepatotoxicity: Treatment-emergent [00:01:00] elevations or worsening of liver tests have occurred. Obtain baseline liver tests and monitor during treatment; monitor also for liver-related adverse reactions and signs of hepatic decompensation. Dose reduction or treatment interruption may be considered if abnormalities occur. Permanently discontinue if persistent liver test abnormalities, clinical hepatitis upon rechallenge, or hepatic decompensation occur.
Gastrointestinal Adverse Reactions: Diarrhea and abdominal pain were reported [00:01:30] as the most common adverse reactions in patients treated with LIVMARLI. Consider reducing the dosage or interrupting treatment if a patient experiences persistent diarrhea or abdominal pain.
Fat-Soluble Vitamin (FSV) Deficiency: Obtain baseline levels and monitor during treatment. Supplement vitamins if deficiency is observed. Consider interrupting LIVMARLI if bone fractures or bleeding occur.
Risk of Propylene Glycol Toxicity (Pediatric Patients Less Than 5 Years of Age): [00:02:00] Total daily intake of propylene glycol should be considered for managing the risk of propylene glycol toxicity. Monitor patients for signs of propylene glycol toxicity. Discontinue LIVMARLI if toxicity is suspected.
[music]
[00:02:18] [END OF AUDIO]
The signs between the fines
Have you spotted any signals of cholestatic pruritus?4,10-12
Sleep disturbances
Irritability
Wiggling
Rubbing the eyes
Digging at the ears
Sleep disturbances
Irritability
Skin damage
Rubbing of the feet
Active scratching
Fidgeting
Unable to focus
Burning, tingling, or prickling sensation
Sleep disturbances
Irritability
Skin damage
Rubbing of the feet
Normalizing the itch
Physical discomfort
Unable to focus
Burning, tingling, or prickling sensation
Start the conversation
Resources to help with the discussion
A guide for health care professionals to help facilitate a better conversation based on normalization phrases you may hear from your patients.
A resource for patients to help identify common signs of cholestatic pruritus, and some that may be misinterpreted or not recognized.



References: 1. Facts about mental health. Mental Health Foundation. Accessed June 11, 2024. https://www.mentalhealth.org.uk/sites/default/files/2022-06/MHF-facts-about-mental-health-posters-colour.pdf 2. Data on file. REF-01349. Mirum Pharmaceuticals, Inc. 3. Data on file. REF-01343. Mirum Pharmaceuticals, Inc. 4. de Azevedo, Takamatsu FY, Kondo M, et al. Pruritus in cholestasis. Rev Soc Bras Clin Med. 2017;15(1):61-67. 5. URSO. Prescribing Information. Axcan Pharma US, Inc. 6. RIFADIN. Prescribing Information. Sanofi-Aventis U.S. LLC. 7. Vistaril. Prescribing Information. Pfizer, Inc. 8. Data on file. REF-01342. Mirum Pharmaceuticals, Inc. 9. Gille S, Griese L, Schaeffer D. Preferences and experiences of people with chronic illness in using different sources of health information: results of a mixed-methods study. Int J Environ Res Public Health. 2021;18(24):13185. doi:10.3390/ijerph182413185 10. Alagille syndrome. MedlinePlus. Updated December 1, 2023. Accessed June 11, 2024. https://medlineplus.gov/genetics/condition/alagille-syndrome 11. Kamath BM, Abetz-Webb L, Kennedy C, et al. Development of a novel tool to assess the impact of itching in pediatric cholestasis. Patient. 2018;11(1):69-82. doi:10.1007/s40271-017-0266-4 12. Elisofon SA, Emerick KM, Sinacore JM, Alonso EM. Health status of patients with Alagille syndrome. J Pediatr Gastroenterol Nutr. 2010;51(6):759-765. doi:10.1097/MPG.0b013e3181ef3771 13. LIVMARLI® (maralixibat) oral solution. Prescribing Information. Mirum Pharmaceuticals, Inc. 14. Gonzales E, Hardikar W, Stormon M, et al. Efficacy and safety of maralixibat treatment in patients with Alagille syndrome and cholestatic pruritus (ICONIC): a randomised phase 2 study. Lancet. 2021;398(10311):1581-1592. doi:10.1016/S0140-6736(21)01256-3 15. Gonzales E, Sturm E, Stormon M, et al. Durability of treatment effect with long-term maralixibat in children with Alagille syndrome: 4-year safety and efficacy. Presented at: American Association for the Study of Liver Diseases Annual Meeting: The Liver Meeting: November 8-12, 2019; Boston, MA. 16. Data on file. REF-00100. Mirum Pharmaceuticals, Inc.
INDICATION
LIVMARLI is indicated for the treatment of cholestatic pruritus in patients who are 3 months of age and older with Alagille syndrome.
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
LIVMARLI is contraindicated in patients with prior or active hepatic decompensation events (eg, variceal hemorrhage, ascites, or hepatic encephalopathy).
WARNINGS AND PRECAUTIONS
Hepatotoxicity: LIVMARLI treatment is associated with a potential for drug-induced liver injury. In the Alagille syndrome trial, treatment-emergent elevations of liver tests or worsening of liver tests occurred.
Obtain baseline liver tests and monitor during treatment. Liver-related adverse reactions and physical signs of hepatic decompensation should also be monitored. Dose reduction or treatment interruption may be considered if abnormalities occur in the absence of other causes. For persistent or recurrent liver test abnormalities, consider treatment discontinuation. Permanently discontinue LIVMARLI if a patient experiences the following: persistent or recurrent liver test abnormalities, clinical hepatitis upon rechallenge, or a hepatic decompensation event.
Gastrointestinal (GI) Adverse Reactions: Diarrhea and abdominal pain were reported as the most common adverse reactions. Monitor for dehydration and treat promptly. Consider reducing the dosage or interrupting LIVMARLI treatment if a patient experiences persistent diarrhea or has diarrhea with bloody stool, vomiting, dehydration requiring treatment, or fever.
Fat-Soluble Vitamin (FSV) Deficiency: Patients can have FSV deficiency (vitamins A, D, E, and K) at baseline, and LIVMARLI may adversely affect absorption of FSVs. Treatment-emergent bone fracture events have been observed more frequently with patients treated with LIVMARLI compared with patients treated with placebo. If bone fractures or bleeding occurs, consider interrupting LIVMARLI and supplement with FSVs. LIVMARLI can be restarted once FSV deficiency is corrected and maintained at corrected levels.
Risk of Propylene Glycol Toxicity (Pediatric Patients Less Than 5 Years of Age): LIVMARLI oral solution contains propylene glycol. Total daily intake of propylene glycol should be considered for managing the risk of propylene glycol toxicity. Monitor patients for signs of propylene glycol toxicity. Discontinue LIVMARLI if toxicity is suspected.
ADVERSE REACTIONS
The most common adverse reactions are diarrhea, abdominal pain, vomiting, FSV deficiency, liver test abnormalities, and bone fractures.
DRUG INTERACTIONS
Administer LIVMARLI at least 4 hours before or 4 hours after administration of bile acid binding resins.
A decrease in the absorption of OATP2B1 substrates (eg, statins) due to OATP2B1 inhibition by LIVMARLI in the GI tract cannot be ruled out. Consider monitoring the drug effects of OATP2B1 substrates as needed.
DOSING INFORMATION
LIVMARLI should be taken 30 minutes before a meal. Refer to the dosing by weight guidelines in the full Prescribing Information for complete details on dosing for the oral solution and tablet formulations. The provided oral solution dosing dispenser must be used to accurately measure the dose. Any remaining LIVMARLI oral solution should be discarded 100 days after first opening the bottle. LIVMARLI tablets can be used in patients weighing ≥25 kg who can swallow tablets.
Please see full Prescribing Information for LIVMARLI.

